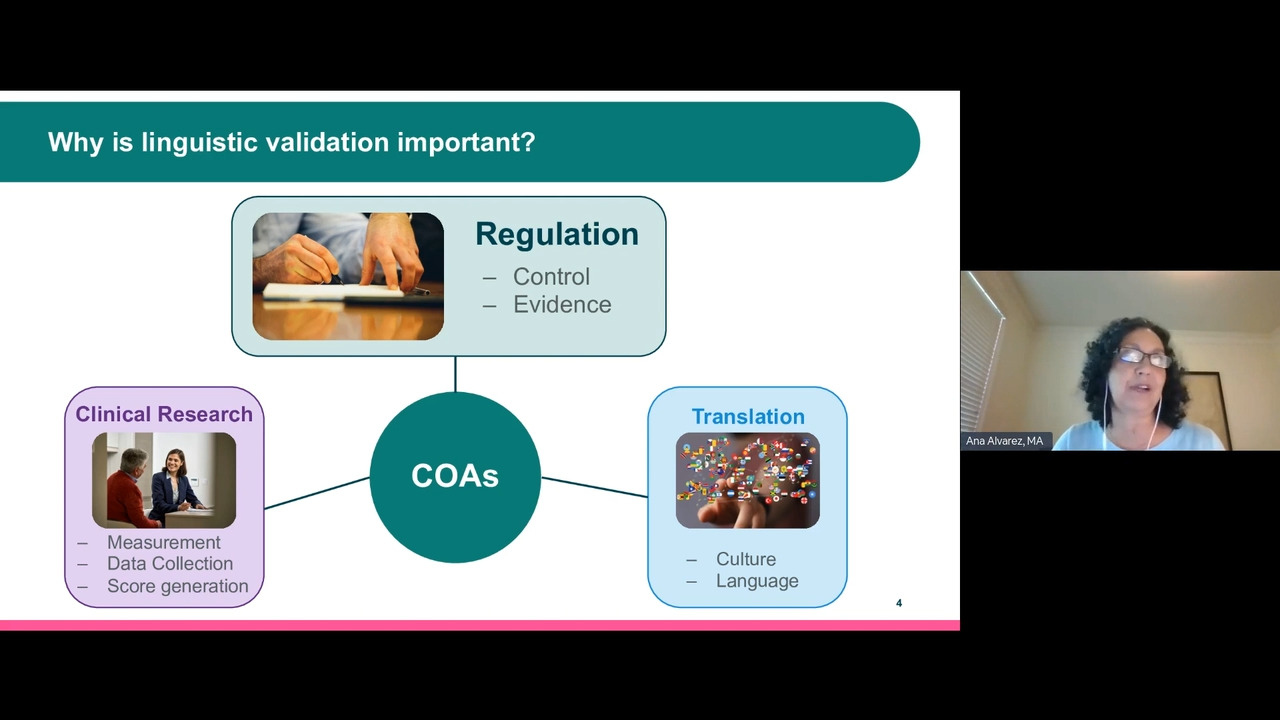
Engagement and retention of patients in clinical trials
44:46
Description
Related Videos
View More
View Less
Related Videos
In Commercialisation & Outcomes
-
Play video Medicare Part D: Price negotiation panel discussion
Medicare Part D: Price negotiation panel discussion
57:55
-
Play video Webinar: Perinatal mood disorders
Webinar: Perinatal mood disorders
51:34
-
Play video Network meta-analysis and its role in evidence-based decision-making
Network meta-analysis and its role in evidence-based decision-making
1:03:09
-
Play video Webinar: Minimising the impact of cross-cultural differences in COA translations
Webinar: Minimising the impact of cross-cultural differences in COA translations
1:01:11
-
Play video Enhancing clinical trials in obesity by integrating COAs and DHTs
Enhancing clinical trials in obesity by integrating COAs and DHTs
58:25
-
Play video Webinar_Strategies to optimise commercialisation of oncology treatments for younger adults
Webinar_Strategies to optimise commercialisation of oncology treatments for younger adults
1:01:45