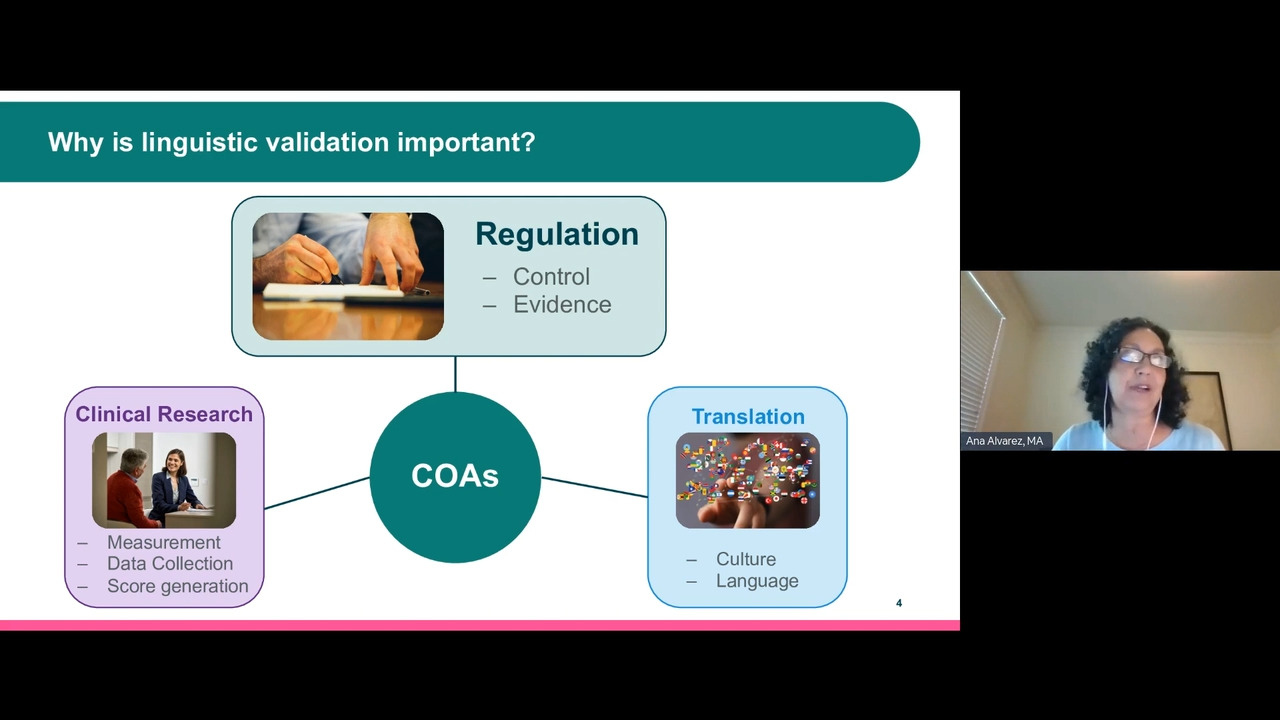
Liguistic Validation of COAS and ECOAS
57:34
Description
Related Videos
View More
View Less
Related Videos
In Commercialisation & Outcomes
-
Play video Webinar: Perinatal mood disorders
Webinar: Perinatal mood disorders
51:34
-
Play video Webinar: Minimising the impact of cross-cultural differences in COA translations
Webinar: Minimising the impact of cross-cultural differences in COA translations
1:01:11
-
Play video Webinar - Regulatory submission writing for NDA sucess
Webinar - Regulatory submission writing for NDA sucess
1:00:10
-
Play video Social media data - Opportunities and insights for Clinical Research
Social media data - Opportunities and insights for Clinical Research
1:01:28
-
Play video Enhancing clinical trials in obesity by integrating COAs and DHTs
Enhancing clinical trials in obesity by integrating COAs and DHTs
58:25
-
Play video Webinar_Strategies to optimise commercialisation of oncology treatments for younger adults
Webinar_Strategies to optimise commercialisation of oncology treatments for younger adults
1:01:45